Technology
Exclusive integrated technology solutions to optimize global clinical supply chains.
Access to data is the key to ensuring clinical trials are managed efficiently. Our integrated suite of technologies have been developed specifically for the clinical trial industry and they provide our Supply Chain Managers with total visibility, enabling them to make faster, informed decisions based on near real-time data. We have integrated the demand forecast, MRP technology and IRT into a single system that provides real-time data. This is completely unique within the clinical supplies industry and again, provides our Supply Chain Managers with a comprehensive, end-to-end view of the supply chain.
Clinical Trial Forecasting and Supply Chain Planning Solutions
SupplyWise™
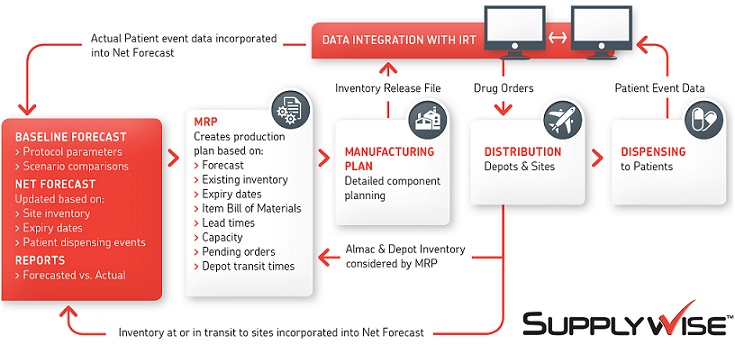
This exciting, innovative system is specifically used by our Supply Chain Managers to proactively manage the supply chain. Our comprehensive solution includes a forecasting tool that calculates clinical supply demand over time, Material Resource Planning (MRP) technology to drive production and capacity planning, and real-time inventory and patient enrollmenT data provided through the Interactive Response Technology (IRT). All of these features, enable the optimzation of production and distribution strategies for efficient clinical supply management.
IRT Solutions
As today’s clinical trial landscape includes studies of all sizes and complexities, our configurable WebEZ® solution, and our fully customisable IXRS™ solution suit all your study needs. We’ll match your study’s protocol functionality with a technological solution that will optimize your trial’s productivity.
Advanced IXRS™
Our interactive voice and web response system offers flexibility and customisability unmatched in the industry. IXRS™ contains configurable and customisable modules, a full suite of languages, alerts, full data integration capabilities and study set-up within weeks of final trial design. When you need the most powerful and flexible tools for patient randomisation and drug supply management, Almac’s IXRS™ is a clear choice.
Almac’s WebEZ™
This web-based, patient randomisation and drug supply management tool offers sponsors a cost-efficient solution for clinical trials. Featuring a user-friendly English interface, easy navigation, standardized reports and a wide range of configuration points, Almac WebEZ® gets your study live within days.
TempEZ™
TempEZ™ offers clients a single database to store temperature data from product manufacture to patient administration, as well as providing a platform to ensure compliance to cGXP and GDP regulations. TempEZ™ is designed to upload temperature data from numerous monitor brands. It also allows any stakeholder in the client’s clinical or commercial supply chain to upload temperature monitor data and record planned and unplanned excursions at a lot or serialized level.

