Software experience & technologies
Almac Diagnostic Services has been delivering custom software solutions for Biopharma clients for many years. The biomarker software team is co-located in the UK and USA.
Our software solutions focus around automation of data processing, filtering targets of interest, quality control assessments, assessing analytical run validity and facilitating patient test reporting outputs. We work closely with clients to identify and refine key requirements at an early stage and tailor our solutions to meet their exact needs, whether this is biomarker software for CTA (Clinical Trial Assays – either RUO or IUO), or CDx (Companion Diagnostics) whilst adhering to appropriate regulatory standards.
We have extensive experience in developing software reporting solutions for quantitative gene expression assays using technologies such as qPCR, nanoString, RNAseq and qualitative DNA assays based on NGS. We also have demonstrable experience in developing reporting software that integrates with upstream analysis pipelines hosted on cloud-based servers.
Regulatory adherence
- Certified compliance with IEC 62304 Medical Device Software: Software life-cycle processes
- Certified compliance with ISO 14971 Risk management for medical devices
- Experience developing software solutions compliant with 21 CFR Part 11 regulations
- Experience in developing software solutions as part of a medical device for regulatory submission e.g. CE marking, FDA approval
- Software solutions developed are validated adhering to Almac Diagnostic Services validation procedures
Software Certifications:
Workflow & processes
Our biomarker software development team have a comprehensive catalog of in-house Standard Operating Procedures (SOPs) which are certified to comply with IEC 62304.
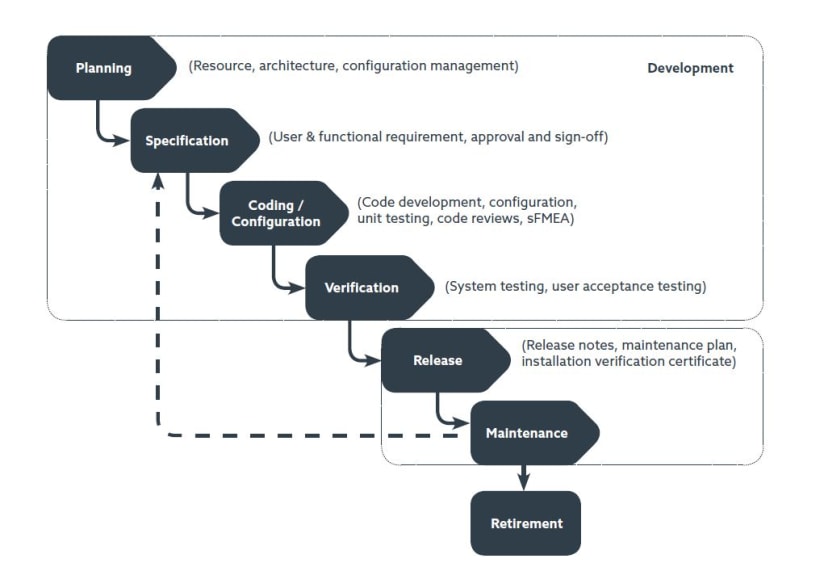
Almac adopts a phased approach to software development and conducts regular design reviews with clients at key stages throughout software development lifecycle.
Almac software process workflow:
Customized software development – Processes compliant with IEC62304.

Find out more. Download the case study:
Almac Diagnostic Services partnered with PredictImmune to develop and validate the gene expression software algorithm used to interpret the results from the world’s first validated and CE-marked prognostic test for Inflammatory Bowel Disease (IBD).
PredictImmune selected Almac as their preferred provider for bioinformatics, software design & development solutions to support the successful commercialization and launch of the PredictSURE IBD™ test.
Click here to download




