Almac Trial Coordinatorᵀᴹ
Fully Integrated eClinical Ecosystem
Almac Trial Coordinator™ is a unified eClinical orchestration solution designed to simplify the execution of complex clinical trials.
It brings together core trial functions including Interactive Response Technology (IRT), electronic Clinical Outcome Assessments (eCOA),
eConsent, visit and participant management, and clinical supply oversight within a single, cohesive ecosystem.
Rather than operating as a collection of disconnected point solutions, Almac Trial Coordinator™ is designed to coordinate workflows,
data, and users across the clinical trial lifecycle. The result is a more efficient, transparent, and site-friendly way to run global studies.
How Almac Trial Coordinatorᵀᴹ Delivers:
Role-based workflows guide next steps, reducing admin, confusion, and quality risks.
Real-time dashboards provide full visibility and control.
Streamlined processes and 24/7 multilingual site support.
Connect seamlessly with IRT, EDC, eCOA, CTMS, and clinical supply systems.
Adapt quickly to complex protocols without compromising quality.
Why Almac Trial Coordinator™?
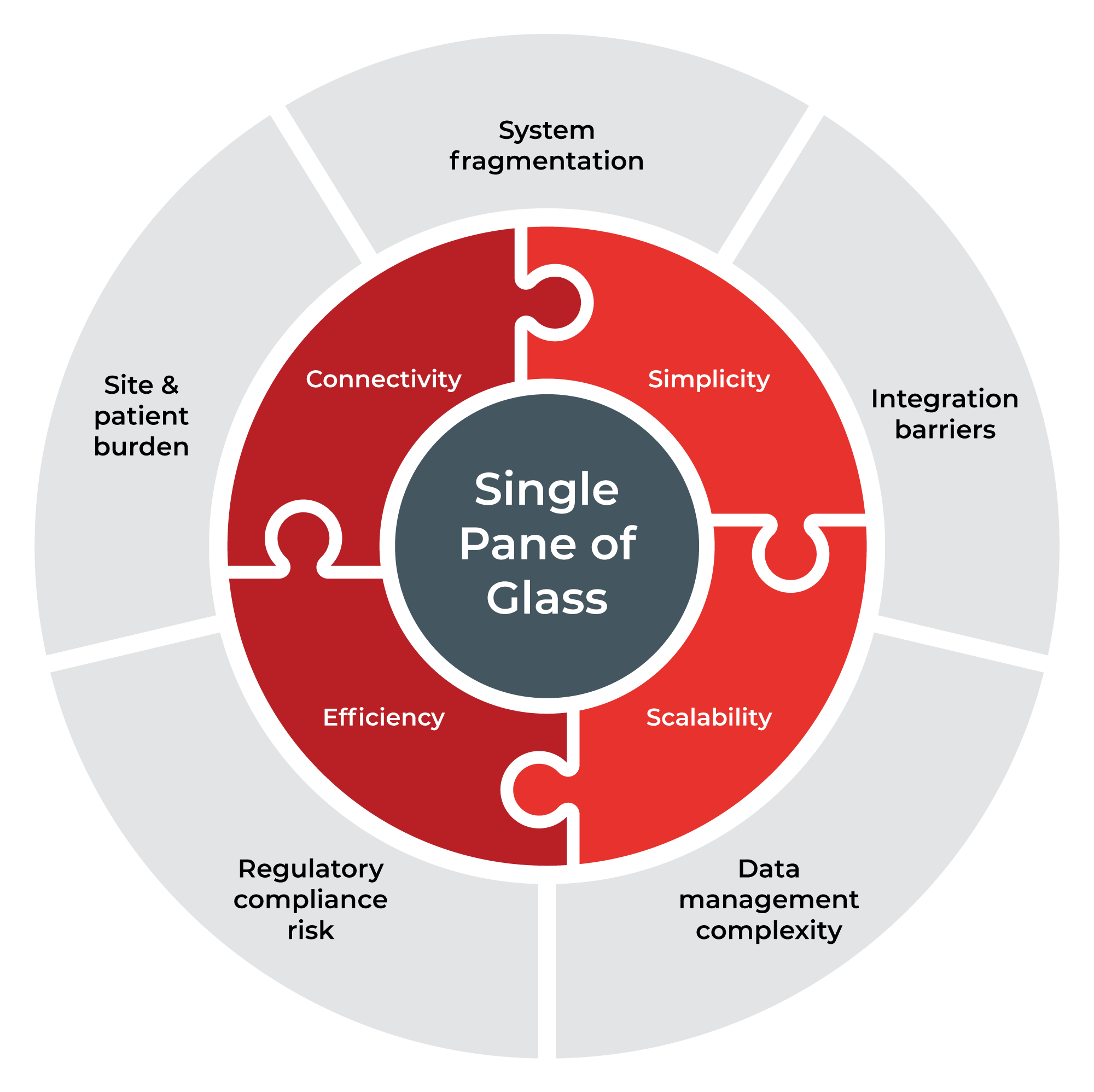
Clinical trials have become increasingly complex, yet the technology supporting them remains fragmented. Sponsors and sites are often required to navigate multiple systems for randomisation, supply management, patient outcomes, consent, and scheduling, each with its own interface, data structure, and support model.
This fragmentation introduces:
- Operational inefficiencies and duplicated effort
- Integration and data reconciliation challenges
- Increased compliance and quality risk
- Significant administrative burden on sites
- Friction in the participant experience
Almac Trial Coordinator™ was created to address these challenges by unifying trial operations into a single orchestrated environment, reducing complexity while maintaining flexibility.
Almac Trial Coordinatorᵀᴹ Key Features
Discover how Almac Trial Coordinatorᵀᴹ transforms clinical trials
IRT, EDC, eCOA, eConsent, and visit management

Workflow management including 3rd party systems
Real-time dashboards for sponsor oversight
Role-based workflows for site enablement
Rapid setup and protocol amendments
24/7 multilingual support
eClinical Solutions for Clinical Trials
Accelerate your clinical trials with our comprehensive eClinical solutions designed for efficiency, compliance, and patient engagement.


