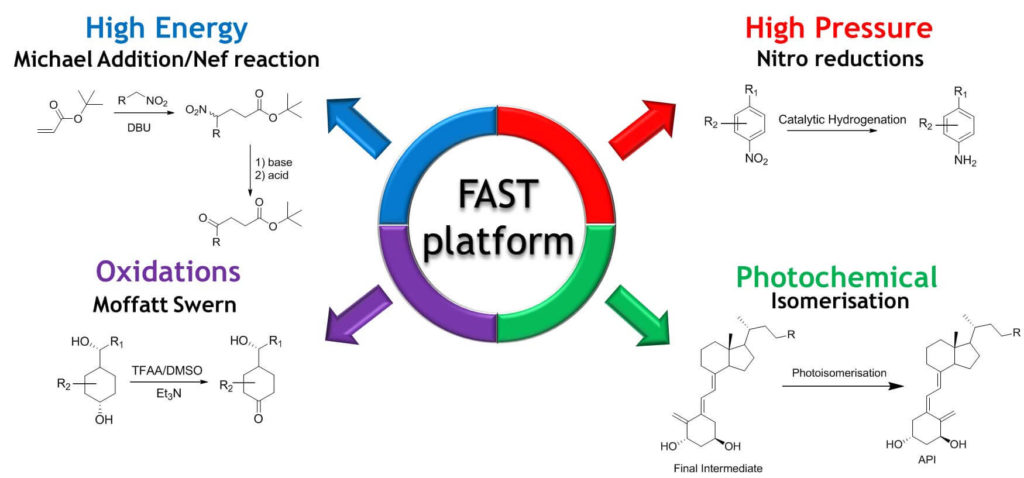
Almac’s Flow Assisted Synthesis Technology platform has developed significant expertise in four key areas:
- High Pressure
- High Energy
- Oxidation
- Photochemistry
Almac continues to drive innovation within their Department of Technology with additional focus on cannabinoids and flow biocatalysis. Multiple grants support our research platform which allows us to undertake high risk but high reward projects including a Knowledge Transfer Partnership and three Horizon 2020 PhD’s with Queen’s University Belfast and a Science Foundation Ireland Industry Fellowship in collaboration with University College Dublin.
Contact us
Case Studies
FAST hydrogenation for aromatic nitro reduction
Our client required development of an existing batch transfer hydrogenation to improve downstream processing.
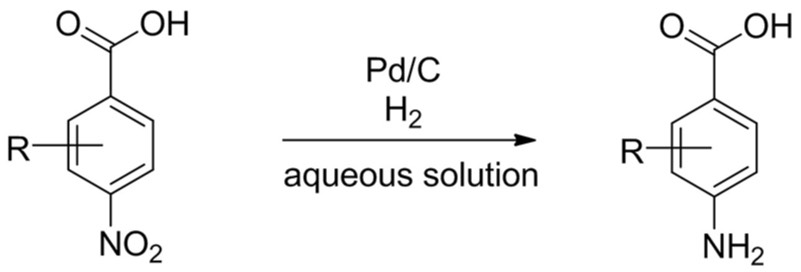
Almac’s solution
Packed bed hydrogenation with rapid screening of in-house pelletised catalyst library, process optimisation and delivery of 1 kg for downstream processing assessment.
Value for the client
Removal of organic solvents from process. Easier product purification due to no catalyst removal issues. Increased control of GTI by-product intermediates.
Michael Addition
Our client required development of a continuous flow process using highly flammable and explosive nitro-alkane building blocks.
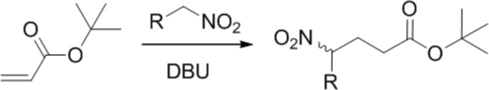
Almac’s solution
Following Almac’s standard workflow under Stage 1 using proof of concept micro-reactor equipment, a continuous flow process demonstrating reaction validity was developed in 4 weeks.
Value for the client
Starting from cheap building blocks and utilising continuous flow accessed advanced chiral intermediates.
Moffatt-Swern oxidation
Our client required a process to access a ketone intermediate by selective oxidation. Batch process required cryogenic temperatures which were not scalable.
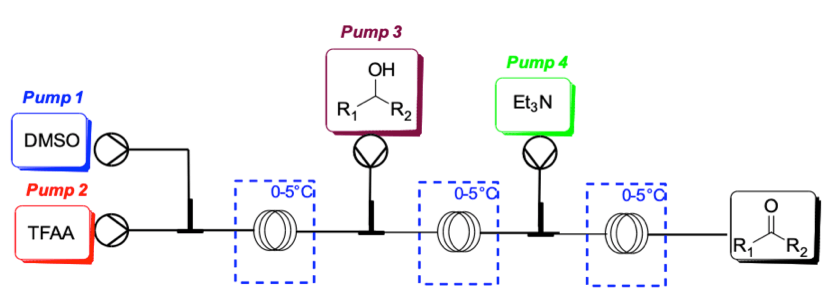
Almac’s solution
Almac took advantage of highly efficient heat transfer in the micro-reactor channels which superseded the limitation of cooling capacity typical of batch vessels. Process was demonstrated at lab scale.
Value for the client
Exploitation of continuous flow circumvented the need for cryogenic temperatures, controlled impurity profile due to less accumulation of activated DMSO and facilitated timely delivery of product.
Photoisomerisation
Our client required delivery of 12 kg of a high potency API where the final step was photoisomerisation under cGMP manufacture.
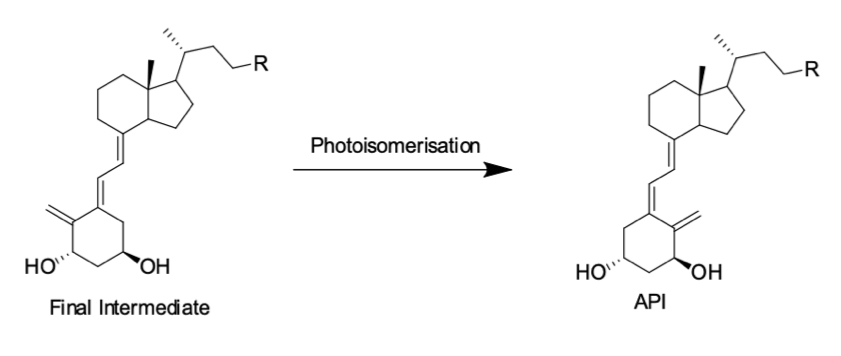
Almac’s solution
Develop a full GMP process with a UV flow through cell to circumvent typical scale up limitations associated with batch photochemistry including screening of filters to set specific wavelengths of light for irradation.
Value for the client
Delivery of API to cGMP manufacture target specification for clinical trials.


