


CDx Manufacturing



CDx Manufacturing



CDx Manufacturing
CDx Manufacturing
Almac Diagnostics Services’ Manufacturing Team has experience in the manufacture of ssPMA lab developed tests and IVD kits. The team regularly engage with the FDA to support pre-submissions, conduct process validation studies, and prepare manufacturing processes for PMA submissions. In addition, the team also provide support to obtain IDE approvals and are instrumental in supporting CE-marking of Clinical Trial Assays.
Almac CDx Manufacturing Capabilities
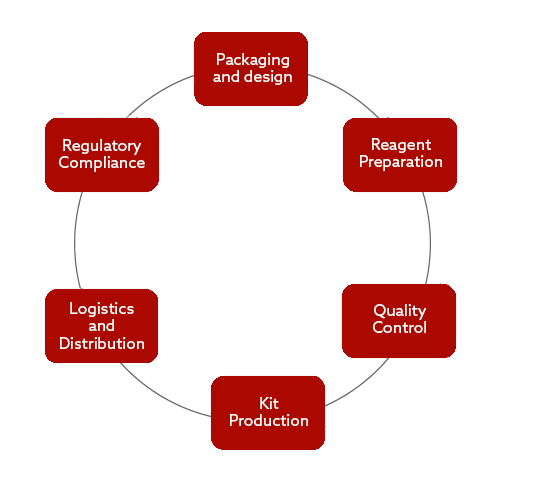
Packaging and Design
- Bespoke packaging & label design service managed by a specialist team
- Labels design to comply with 21CFR 801/809 and ISO15223
- Label material validation
- Technical consultation for component selection
- Inventory and temperature-controlled solutions for stock
Reagent Preparation
- Dedicated suite for the preparation of reagents and control samples
- Environmentally monitored facilities
- Robust QC procedures
Quality Control
- Development and validation of QC test methods
- Validation of the Manufacturing Process
- Dedicated equipment lines
- Sampling and component testing
- Manufacturing process surveillance
Kit Production
- Set up and assembly of dedicated production lines for a range of IVD kits and batch sizes
- Inventory management system for stock monitoring
- Temperature controlled facilities and warehouse
- Controlled production processes and robust staff training
Logistics and Distribution
- Dedicated logistics team to provide solutions for IVD kit distribution
- Approved couriers and worldwide delivery capabilities
- Bespoke distribution strategy per product
- Partners with global distributors
- Cold chain specialists
Regulatory Compliance
- GMP compliant and ISO13485 accredited facility
- Independent quality and regulatory personnel
- Facilitation of customer / regulatory audits
- Regulatory compliant labelling
