US Workshops: Considerations for EU Product Launch
Location
- Starts: 7 June 2026
- Ends: 7 June 2026
Launching your product on to the European Market can present a real challenge if you are not familiar with the various country specific requirements. With its current 28 member states, numerous official working languages and complex regulatory framework, this workshop will clarify the key requirements for a successful EU product launch and will discuss the impact Brexit could have on your launch strategy and future supply chain.
Helping you to be better prepared for what lies ahead and ensure your commercial launch is successful, our subject matter experts will guide you through the EU launch process, from submitting your MAA, to final distribution of drug product to the end-user.
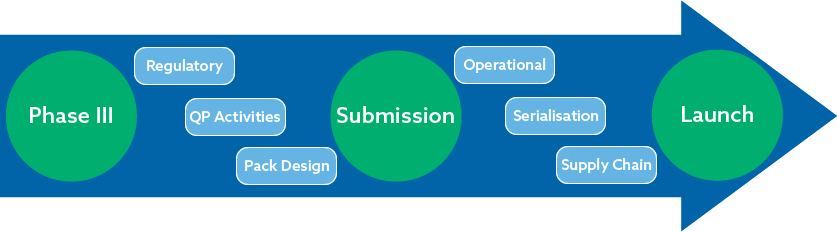
Key workshop objectives
- Learn about the European regulatory framework and how this determines all your launch milestones
- Understand the importance of the Qualified Person (QP) and QP declarations
- Discover key EU packaging considerations
- Gain insights into managing a complex supply chain
- Learn about the key Serialisation readiness requirements when partnering with a CMO
- Identify the top operational considerations as you progress to commercialisation
Roundtable discussions
Not only will our experts share their product launch knowledge and experience but they will be hosting specialist roundtable discussions to address your specific product launch queries.
These roundtable discussions will give you the opportunity to speak with our subject matter experts and ask them more specific questions relevant to your particular project.