Rational Design of a (S)-Selective-Transaminase for Asymmetric Synthesis of (1S)-1-(1,1′-biphenyl-2-yl)ethanamine
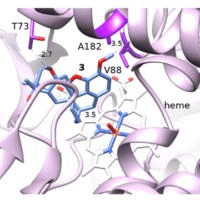
Amine transaminases offer an environmentally sustainable synthesis route for the production of pure chiral amines. However, their catalytic efficiency toward bulky ketone substrates is greatly limited by steric hindrance and therefore presents a great challenge for industrial synthetic applications.We hereby report an example of rational transaminase enzyme design to help alleviate these challenges.
Starting from the Vibrio fluvialis amine transaminase that has no detectable catalytic activity toward the bulky aromatic ketone 2-acetylbiphenyl, we employed a rational design strategy combining in silico and in vitro studies to engineer the transaminase enzyme with a minimal number of mutations, achieving an high catalytic activity and high enantioselectivity. We found that, by introducing two mutations W57G/R415A, detectable enzyme activity was achieved.