UAT: A Custom-Built Roadmap to Kick Off Your Clinical Trial Journey
By Antoinette Walsh
Director of Technical Solutions
What is the purpose of UAT?
The purpose of a UAT is to ensure the software, in this case IRT, was built to the specifications. Sponsors have a regulatory responsibility to perform UAT as outlined in EMA GCP 600788, Section 2.2.4.
UAT is your opportunity to get your hands on your custom-built solution to ensure it will meet your end users’ needs as you envisioned. This is not the time to ”Break the System” (your vendor team has likely already performed this), but you do want to run through transactions as your end-user would typically. You will want to ensure not only are the transactions completing as expected, but the associated reporting and data exports are accurate. If you do find any issues, UAT is a more cost-efficient check point to correct them prior to production use.
What are the keys to a successful UAT?
You will have limited time to execute your UAT and so you need to make sure you have a plan of action from the beginning of your project. You will also want to ensure your other vendor’s IRT integrated systems are ready at the same time to ensure your data is integrating across systems as expected, mirroring how the system will be utilized in production end-to-end. Partnering with a vendor with expertise on the UAT script creation process can help to streamline many of the activities that happen during this time.

Plan
Create a project plan: The IRT is just one system for your clinical trial. A best practice is to ensure your integrated systems are available in a UAT environment at the same time as your IRT. This will help you validate test files as you execute IRT transactions
Create your Subject Matter (SME) team: who will provide input and approval to the UAT strategy document and UAT test scripts? Who will help execute the scripts? What design enhancements or changes are dealbreakers to closing the UAT Phase? Who needs to give final approval to move the system Live?
Prepare
Review the software specifications and prepare the UAT testing scenarios you plan to cover during the software build in a UAT strategy document. The document should be a high-level list of key scenarios that must be covered in the plan. Once you have identified your strategy, get feedback on the UAT approach from your stakeholders including internal SME’s and external vendors. When all parties have reviewed and requirements are stable, you can draft your formal scripts off this scenario document.
Your test scripts should focus on Critical/High-Risk scenarios. Leverage your vendor’s core and configurable functionality to limit your testing to the customized areas of the software. A great place to bring efficiency into your script is to re-use data created earlier in the script for later transactions. For example, you can screen a subject and carry through Randomization, Kit Assignment Visits, and End of Treatment rather than using new subjects for each feature. A step further for more advanced UAT teams to consider is using test automation tooling to speed up repetitive processes such as subject data setup.
How will you execute your scripts?
There are several ways a UAT can be executed. Here are a few we recommend and considerations you may want to think about.
- Independently: One user executes all scripts
- Team based: Will the team use the same script or different scripts? What methodology will be followed? Here are a few types of team-based UAT processes we have seen in our experience:
UAT Methodologies
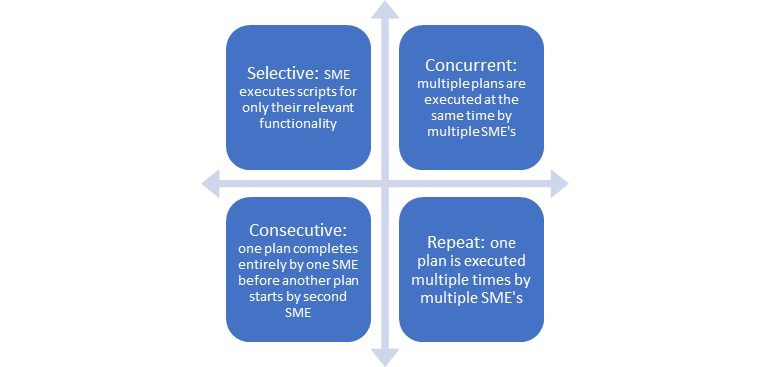

How will the team collaborate and communicate questions/challenges/concerns?
How technology can be harnessed to bring together virtual/global teams?
Perform
The clock is ticking! Execute your scripts and add extra ones as you think of more you missed. Communicate status with key stakeholders each day in summary form. Keep a log of issues/questions and close out any showstopper issues with the vendor prior to UAT completion. Defer any non-priority changes to a later version of the software as time allows.
Approve
When all parties have completed their tests and agree that the system was built to the approved requirements, you can now give approval to the vendor declaring the acceptability of the results and request that the system or change be moved to the live environment. Congratulations! You have successfully completed the UAT Phase and meeting your regulatory obligations to ensure suitability of the system!
Final Thoughts
User Acceptance Testing is a software process that gives sponsors the opportunity to run through real-world transactions prior to system launch. UAT functions as a cost-effective checkpoint that ensures efficiency, suitability, and end-user performance as envisioned.





