Biocatalytic dearomatisation of para-fluorobenzoic acid
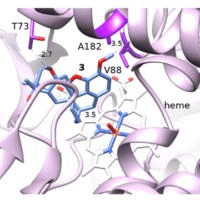
The enzyme benzoate dioxygenase (BZDO) from Ralstonia eutropha B9 is able to dihydroxylate benzoic acids in a dearomative process that proceeds with a different regioselectivity than other known dioxygenase enzymes.
Here we show that 4-fluorobenzoic acid is oxidized by BZDO to give an enantiopure diol that can be rapidly elaborated to highly oxygenated homochiral building blocks with quaternary centers. Notably, the diol produced in this biotransformation displays reactivity which is distinct from that of the more extensively studied non-fluorinated analog.