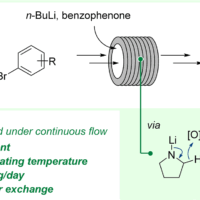
Crystallization Process Development
As a world leader in pharmaceutical services, Almac delivers bespoke crystallization development work programs for small molecules and peptides designed to provide a robust, scaleable, and transferrable crystallization procedure to help accelerate clinical development and minimize risk in any downstream activities by eliminating batch-to-batch variability. The goals of typical studies include:
- Impurity rejection / purge studies
- Kinetics of nucleation and growth
- Polymorphic form control
- Maximizing yield
- Morphology engineering studies
- Particle size control
- Bulk density and powder flow improvement
- Milling and micronisation studies
- Isolation and drying processes
- Control over residual solvent
- Seeding protocol
Read the full article below to learn more.
View Resource