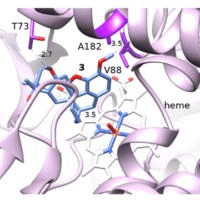
Mechanism of Phosphatidylglycerol Activation Catalyzed by Prolipoprotein Diacylglyceryl Transferase
Lipoproteins are essential for bacterial survival. Bacterial lipoprotein biosynthesis is accomplished by sequential modification by three enzymes in the inner membrane, all of which are emerging antimicrobial targets.The X-ray crystal structure of prolipoprotein diacylglyceryl transferase (Lgt) and apolipoprotein N-acyl transferase (Lnt) has been reported.
However, the mechanisms of the posttranslational modification catalyzed by these enzymes have not been understood. Here, we studied the mechanism of the transacylation reaction catalyzed by Lgt, the first enzyme for lipoprotein modification using molecular docking, molecular dynamics, and quantum mechanics/molecular mechanics (QM/MM) calculations.
Published in the Journal of Physical Chemistry